Which of the Compounds Can Exist as Optical Isomers (Enantiomers)?
STEREOISOMERISM - OPTICAL ISOMERISM
Optical isomerism is a course of stereoisomerism. This page explains what stereoisomers are and how you recognise the possibility of optical isomers in a molecule.
What is stereoisomerism?
What are isomers?
Isomers are molecules that take the same molecular formula, just have a dissimilar arrangement of the atoms in space. That excludes any different arrangements which are merely due to the molecule rotating as a whole, or rotating nearly detail bonds.
Where the atoms making upwards the various isomers are joined upwards in a different social club, this is known as structural isomerism. Structural isomerism is not a course of stereoisomerism, and is dealt with on a separate folio.
Note:If you aren't sure nearly structural isomerism, it might be worth reading about information technology before yous proceed with this page.
What are stereoisomers?
In stereoisomerism, the atoms making upwardly the isomers are joined up in the aforementioned order, but still manage to have a different spatial arrangement. Optical isomerism is ane form of stereoisomerism.
Optical isomerism
Why optical isomers?
Optical isomers are named similar this considering of their result on airplane polarised light.
Help!If you don't understand about plane polarised light, follow this link earlier you go on with this page.
Simple substances which bear witness optical isomerism exist as two isomers known as enantiomers .
-
A solution of one enantiomer rotates the aeroplane of polarisation in a clockwise management. This enantiomer is known as the (+) form.
For example, one of the optical isomers (enantiomers) of the amino acrid alanine is known every bit (+)alanine.
-
A solution of the other enantiomer rotates the plane of polarisation in an anti-clockwise management. This enantiomer is known equally the (-) form. And so the other enantiomer of alanine is known as or (-)alanine.
-
If the solutions are every bit full-bodied the amount of rotation acquired by the two isomers is exactly the same - only in opposite directions.
-
When optically agile substances are made in the lab, they often occur as a 50/fifty mixture of the two enantiomers. This is known as a racemic mixture or racemate . It has no effect on plane polarised calorie-free.
Note:One of the worrying things about optical isomerism is the number of obscure words that suddenly get thrown at you. Bear with it - things are soon going to get more than visual!
At that place is an alternative way of describing the (+) and (-) forms which is potentially very disruptive. This involves the employ of the lowercase messages d- and l-, standing for dextrorotatory and laevorotatory respectively. Unfortunately, there is another different use of the capital letters D- and L- in this topic. This is totally confusing! Stick with (+) and (-).
How optical isomers arise
The examples of organic optical isomers required at A' level all comprise a carbon cantlet joined to iv different groups. These two models each accept the same groups joined to the central carbon atom, but still manage to be different:
Obviously as they are drawn, the orange and blue groups aren't aligned the same style. Could you get them to align by rotating one of the molecules? The side by side diagram shows what happens if yous rotate molecule B.
They still aren't the same - and there is no fashion that you can rotate them so that they look exactly the aforementioned. These are isomers of each other.
They are described as being not-superimposable in the sense that (if you imagine molecule B beingness turned into a ghostly version of itself) you couldn't slide one molecule exactly over the other one. Something would always be pointing in the wrong management.
Annotation:Unless your visual imagination is reasonably good, this is much easier to understand if you have actually got some models to play with. If your school or higher hasn't given you lot the opportunity to play around with molecular models in the early stages of your organic chemistry form, you might consider getting agree of a cheap gear up. The models made by Molymod are both cheap and easy to use. An introductory organic set is more than adequate. Google molymod to observe a supplier and more about them, or take a look at this set up or this set or something similar from Amazon.
Share the cost with some friends, go on it in good condition and don't lose any $.25, and resell information technology via eBay or Amazon at the finish of your grade.
Alternatively, go hold of some coloured Plasticene (or other children'due south modelling clay) and some used matches and brand your ain. It'southward cheaper, but more hard to get the bond angles right.
What happens if two of the groups attached to the central carbon atom are the same? The next diagram shows this possibility.
The ii models are aligned exactly equally before, only the orange grouping has been replaced by another pinkish 1.
Rotating molecule B this fourth dimension shows that it is exactly the same as molecule A. Yous only get optical isomers if all iv groups attached to the primal carbon are different.
Chiral and achiral molecules
The essential divergence betwixt the two examples we've looked at lies in the symmetry of the molecules.
If there are two groups the same attached to the central carbon atom, the molecule has a airplane of symmetry. If yous imagine slicing through the molecule, the left-manus side is an exact reflection of the right-hand side.
Where there are iv groups attached, there is no symmetry anywhere in the molecule.
A molecule which has no aeroplane of symmetry is described as chiral . The carbon atom with the four different groups attached which causes this lack of symmetry is described equally a chiral centre or equally an asymmetric carbon atom .
The molecule on the left higher up (with a plane of symmetry) is described as achiral .
Only chiral molecules have optical isomers.
The relationship between the enantiomers
One of the enantiomers is simply a non-superimposable mirror image of the other i.
In other words, if one isomer looked in a mirror, what information technology would encounter is the other one. The two isomers (the original i and its mirror paradigm) have a different spatial arrangement, and so can't be superimposed on each other.
If an achiral molecule (one with a aeroplane of symmetry) looked in a mirror, you would always find that by rotating the prototype in infinite, you could make the two look identical. It would be possible to superimpose the original molecule and its mirror epitome.
Some real examples of optical isomers
Butan-ii-ol
The asymmetric carbon cantlet in a compound (the ane with 4 different groups fastened) is often shown past a star.
It'southward extremely important to draw the isomers correctly. Draw one of them using standard bond notation to show the 3-dimensional arrangement around the asymmetric carbon atom. And so draw the mirror to show the examiner that you know what you are doing, and and then the mirror image.
Assistance!If you don't empathize this bond notation, follow this link to drawing organic molecules earlier you keep with this folio.
Notice that you don't literally draw the mirror images of all the letters and numbers! It is, withal, quite useful to contrary large groups - expect, for example, at the ethyl grouping at the top of the diagram.
It doesn't matter in the least in what club y'all draw the four groups around the cardinal carbon. Equally long equally your mirror paradigm is drawn accurately, you will automatically have drawn the two isomers.
So which of these two isomers is (+)butan-2-ol and which is (-)butan-2-ol? At that place is no simple way of telling that. For A'level purposes, you tin merely ignore that trouble - all you need to be able to do is to draw the two isomers correctly.
two-hydroxypropanoic acid (lactic acrid)
Once again the chiral centre is shown by a star.
The ii enantiomers are:
It is important this time to draw the COOH group backwards in the mirror image. If you don't in that location is a good chance of you joining it on to the primal carbon wrongly.
If you depict it similar this in an examination, you won't get the mark for that isomer even if you have drawn everything else perfectly.
2-aminopropanoic acid (alanine)
This is typical of naturally-occurring amino acids. Structurally, it is just similar the last example, except that the -OH group is replaced by -NH2
The two enantiomers are:
Only one of these isomers occurs naturally: the (+) class. You can't tell but by looking at the structures which this is.
Information technology has, however, been possible to work out which of these structures is which. Naturally occurring alanine is the right-hand structure, and the way the groups are arranged around the central carbon atom is known as an L- configuration. Discover the apply of the capital L. The other configuration is known as D-.
So you lot may well observe alanine described as L-(+)alanine .
That ways that it has this detail construction and rotates the airplane of polarisation clockwise.
Even if yous know that a different compound has an arrangement of groups similar to alanine, yous still tin can't say which way it will rotate the aeroplane of polarisation.
The other amino acids, for instance, have the same arrangement of groups as alanine does (all that changes is the CH3 grouping), but some are (+) forms and others are (-) forms.
It's quite mutual for natural systems to only work with one of the enantiomers of an optically active substance. Information technology isn't too difficult to encounter why that might be. Because the molecules accept different spatial arrangements of their various groups, only one of them is likely to fit properly into the active sites on the enzymes they piece of work with.
In the lab, it is quite common to produce equal amounts of both forms of a chemical compound when it is synthesised. This happens just by chance, and you tend to get racemic mixtures.
Identifying chiral centres in skeletal formulae
A skeletal formula is the virtually stripped-downwards formula possible. Look at the structural formula and skeletal formula for butan-two-ol.
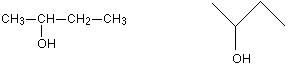
Find that in the skeletal formula all of the carbon atoms accept been left out, as well equally all of the hydrogen atoms attached to carbons.
In a skeletal diagram of this sort:
-
there is a carbon atom at each junction between bonds in a chain and at the cease of each bond (unless in that location is something else there already - like the -OH group in the case);
-
in that location are enough hydrogen atoms fastened to each carbon to make the total number of bonds on that carbon up to 4.
We have already discussed the butan-2-ol case further up the page, and y'all know that it has optical isomers. The second carbon atom (the one with the -OH attached) has iv different groups around it, and so is a chiral center.
Is this obvious from the skeletal formula?
Well, it is, provided you remember that each carbon atom has to have 4 bonds going abroad from it. Since the second carbon here only seems to have 3, at that place must also be a hydrogen attached to that carbon. So information technology has a hydrogen, an -OH grouping, and 2 different hydrocarbon groups (methyl and ethyl).
Four different groups effectually a carbon atom means that it is a chiral centre.
A slightly more complicated case: two,iii-dimethylpentane
The diagrams show an uncluttered skeletal formula, and a repeat of it with 2 of the carbons labelled.

Look beginning at the carbon atom labelled two. Is this a chiral heart?
No, it isn't. Two bonds (one vertical and ane to the left) are both attached to methyl groups. In addition, of course, there is a hydrogen cantlet and the more complicated hydrocarbon group to the right. It doesn't have iv different groups attached, and and then isn't a chiral centre.
What about the number 3 carbon atom?
This has a methyl group below information technology, an ethyl grouping to the right, and a more complicated hydrocarbon grouping to the left. Plus, of course, a hydrogen atom to brand up the 4 bonds that take to be formed by the carbon. That means that it is attached to 4 dissimilar things, so is a chiral eye.
Introducing rings - further complications
At the time of writing, ane of the UK-based examination boards (Cambridge International - CIE) normally asked most the number of chiral centres in some very complicated molecules involving rings of carbon atoms. The rest of this page is to teach you how to cope with these.
We will first with a fairly simple ring chemical compound:
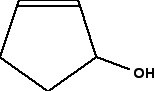
When you are looking at rings like this, as far as optical isomerism is concerned, you lot don't demand to look at whatever carbon in a double bond. You also don't demand to look at any junction which only has 2 bonds going abroad from it. In that case, there must exist 2 hydrogens fastened, and so there tin't possibly be four unlike groups fastened.
In this instance, that ways that you but demand to look at the carbon with the -OH group attached.
It has an -OH group, a hydrogen (to brand upward the total number of bonds to four), and links to ii carbon atoms. How does the fact that these carbon atoms are part of a ring affect things?
Y'all but need to trace back effectually the band from both sides of the carbon you are looking at. Is the organisation in both directions exactly the aforementioned? In this case, it isn't. Going in 1 direction, you come immediately to a carbon with a double bond. In the other direction, you run across two singly bonded carbon atoms, and then i with a double bond.
That means that y'all oasis't got two identical hydrocarbon groups attached to the carbon yous are interested in, and then it has four unlike groups in total around it. It is asymmetric - a chiral centre.
What nearly this near-relative of the last molecule?
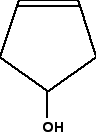
In this example, everything is as before, except that if you lot trace around the ring clockwise and anticlockwise from the carbon at the bottom of the ring, in that location is an identical pattern in both directions. You can think of the bottom carbon beingness attached to a hydrogen, an -OH group, and two identical hydrocarbon groups.
It therefore isn't a chiral centre.
The other affair which is very noticeable nearly this molecule is that there is a plane of symmetry through the carbon atom we are interested in. If you chopped it in half through this carbon, ane side of the molecule would be an exact reflection of the other. In the first ring molecule above, that isn't the example.
If you tin can see a plane of symmetry through the carbon atom it won't be a chiral centre. If there isn't a plane of symmetry, it volition be a chiral centre.
A seriously complicated case - cholesterol
The skeletal diagram shows the structure of cholesterol. Some of the carbon atoms have been numbered for discussion purposes below. These are not function of the normal system for numbering the carbon atoms in cholesterol.
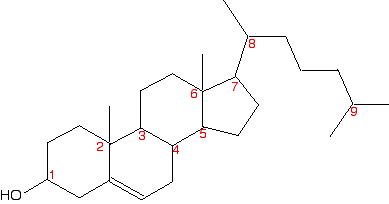
Before yous read on, expect advisedly at each of the numbered carbon atoms, and determine which of them are chiral centres. The other carbon atoms in the structure tin can't exist chiral centres, because they are either parts of double bonds, or are joined to either two or three hydrogen atoms.
Notation:I am being deliberately unkind hither! Ordinarily when a molecule like cholesterol is discussed in this context, extra detail is often added to the skeletal construction. For example, important hydrogen atoms or methyl groups are drawn in. It is salubrious to have to do it the hard way!
So . . . how many chiral centres did yous find? In fact, there are 8 chiral centres out of the total of 9 carbons marked. If you didn't notice all eight, go back and have another look before you lot read any further. It might help to sketch the construction on a piece of newspaper and depict in any missing hydrogens attached to the numbered carbons, and write in the methyl groups at the cease of the branches as well.
This is done for you below, only it would be a lot meliorate if you did it yourself and then checked your sketch later.
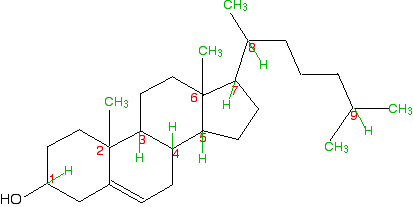
Starting with the easy one - information technology is obvious that carbon ix has 2 methyl groups attached. It doesn't take four unlike groups, and then can't be chiral.
If y'all take a general look at the residual, information technology is fairly clear that none of them has a aeroplane of symmetry through the numbered carbons. Therefore they are all probable to exist chiral centres. But it's worth checking to run across what is fastened to each of them.
Carbon ane has a hydrogen, an -OH and two unlike hydrocarbon chains (really bits of rings) fastened. Bank check clockwise and anticlockwise, and yous will run into that the arrangement isn't identical in each direction. 4 dissimilar groups means a chiral centre.
Carbon 2 has a methyl and iii other unlike hydrocarbon groups. If you bank check along all three bits of rings , they are all different - another chiral centre. This is besides true of carbon 6.
Carbons iii, 4, 5 and vii are all basically the same. Each is attached to a hydrogen and three different bits of rings. All of these are chiral centres.
Finally, carbon viii has a hydrogen, a methyl grouping, and two different hydrocarbon groups fastened. Once more, this is a chiral center.
This all looks difficult at first glance, but information technology isn't. You do, however, have to take a nifty deal of care in working through information technology - information technology is amazingly easy to miss 1 out.
-
Where would you similar to become now?
-
To the isomerism carte du jour. . .
To carte of basic organic chemical science. . .
To Main Menu . . .
![]()
© Jim Clark 2000 (concluding modified November 2012)
Source: https://www.chemguide.co.uk/basicorg/isomerism/optical.html
Postar um comentário for "Which of the Compounds Can Exist as Optical Isomers (Enantiomers)?"